OSAKA, TOKYO and IWATE, June 30, 2022 – Nitto Denko Corporation (TSE: 6988, President: Hideo Takasaki, “Nitto”), Astellas Pharma Inc. (TSE: 4503, President and CEO: Kenji Yasukawa, Ph.D., “Astellas” ), and M. Heart Co., Ltd. (President and CEO: Yoshimi Mizunuma, “M. Heart”) today announced that the three companies have entered into an agreement for a sales pilot of disposable Holter ECG device "EG HolterTM" for the Japan market.
Based on this agreement, Astellas has established an e-commerce site for healthcare professionals and will start the sales pilot of the device today. After verifying the business model through the sales pilot, Astellas will move to full-scale marketing of the product.
EG Holter is a holter electrocardiograph designed and developed by Nitto. Because it is a disposable device, it is hygienic and does not require maintenance. It is 6 mm thick,11 g in weight, has no cords, and is water resistance (IPX4). It is easy to attach and remove.
Data obtained by EG Holter will be analyzed by "MYHOLTER II", which is software for the holter analyzer jointly developed by Astellas and M. Heart. MYHOLTER II is a program that analyzes holter ECG data with a proprietary analytical algorithm using artificial intelligence (AI). M. Heart has obtained cerification for EG Holter and MYHOLTER II as medical device (Class II).
Nitto, Astellas, and M. Heart have come together to create a simple, convenient, and patient-centered solution for patients and medical institutions.
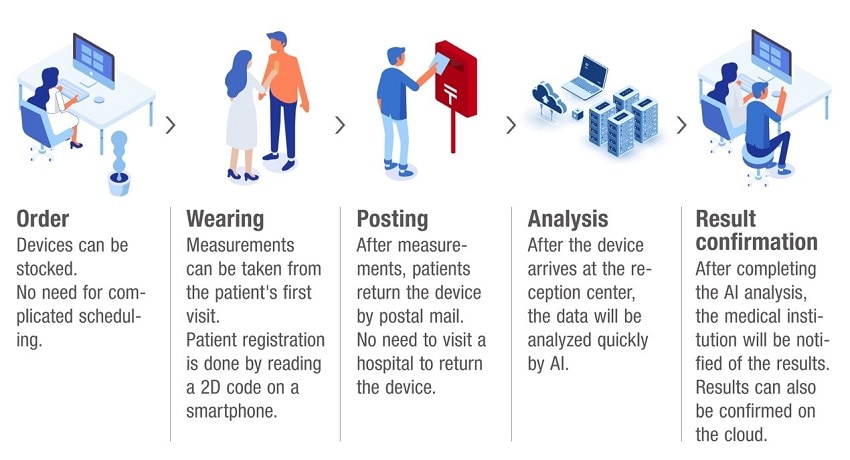 Analysis flow using EG Holter and MYHOLTER II |
Medical costs for heart diseases and other cardiovascular diseases in Japan exceed 6 trillion yen per year―the highest of any disease category1. Of particular concern is cardiogenic cerebral embolism, which occurs when a thrombus that formed in the heart blocks an artery in the brain or neck. This has a high mortality rate (20%) and often results in severe sequelae, such as a bedridden state (40%)2. Atrial fibrillation is said to be the cause of cardiogenic cerebral embolism in 3 out of 4 cases2. Early detection of atrial fibrillation is an important social issue.
By providing a total solution that involves combining convenient ECG testing using the EG Holter and the data analysis using MYHOLTER II, Nitto, Astellas, and M. Heart propose to enable early detection and appropriate treatment of atrial fibrillation, a condition estimated to affect approximately 700,000 patients in Japan2, and contribute to the prolongation of healthy life expectancy.
About Nitto
Nitto is a Japanese manufacturer of high-performance materials. Since its founding in 1918, they provide a wide range of products based on four core technologies: adhesion, coating, polymer function control, and polymer analysis & evaluation. A few examples are polarizing film, indispensable for displays of smartphones and TVs, industrial adhesive tape, automobile parts and medical supplies. Remaining faithful to the brand slogan, “Innovation for Customers,” Nitto continues to contribute to a better society and environment through its innovative products and services and delivers technologies that shape the future. For further information, please visit Nitto’s official website.
About Astellas
Astellas Pharma Inc. is a pharmaceutical company conducting business in more than 70 countries around the world. We are promoting the Focus Area Approach that is designed to identify opportunities for the continuous creation of new drugs to address diseases with high unmet medical needs by focusing on Biology and Modality. Furthermore, we are also looking beyond our foundational Rx focus to create Rx+® healthcare solutions that combine our expertise and knowledge with cutting-edge technology in different fields of external partners. Through these efforts, Astellas stands on the forefront of healthcare change to turn innovative science into value for patients. For more information, please visit our website at https://www.astellas.com/en.
About M. Heart
M. Heart is a MedTech venture that provides the cloud-type holter ECG analysis service to medical institutions nationwide using the program “My Holter”.
Since the data to be analyzed complies with the international standard MFER, it enables collaboration with domestic and overseas electrocardiograph manufacturers and system manufacturers. Our staff includes doctors, clinical laboratory engineers, IT engineers, marketers, and pharmaceutical consultants, and we will realize business transformation of medical services with innovative technology and diverse imagination.
For more information on M-Heart, please visit our website at https://www.emheart.co.jp/
Cautionary Notes
In this press release, statements made with respect to current plans, estimates, strategies and beliefs and other statements that are not historical facts are forward-looking statements about the future performance of Nitto. These statements are based on management’s current assumptions and beliefs in light of the information currently available to it and involve known and unknown risks and uncertainties. A number of factors could cause actual results to differ materially from those discussed in the forward-looking statements. Such factors include, but are not limited to: (i) changes in general economic conditions and in laws and regulations, (ii) currency exchange rate fluctuations, (iii) delays in new product launches, (iv) the inability of Nitto to market existing and new products effectively, (v) the inability of Nitto to continue to effectively create solutions accepted by customers in highly competitive markets, and (vi) infringements of Nitto’ intellectual property rights by third parties. Information about medical devices (including products currently in development) which is included in this press release is not intended to constitute an advertisement or medical advice.
Nitto Denko Corporation
Brand Strategy Dept.
TEL: +81-6-7632-2101 FAX: +81-6-7632-2568
Astellas Pharma Inc.
Corporate Advocacy & Relations
TEL: +81-3-3244-3201
M.Heart Co,ltd
https://www.emheart.co.jp/contact/
1: Ministry of Health, Labour and Welfare, 2019 Summary of National Medical Care Expenditures
2: From Japan Stroke Association, Japanese Heart Rhythm Society, Atrial Fibrillation Weekly Website
For any inquiries about this press release.
Business Hours (EST)
8:00 a.m.-5:30 p.m.(Except for Sat, Sun, and Holidays)